A Comparative Study of the Adsorption of Industrial Dyes onto Grape Cores-Based Adsorbents from Aqueous Media
Keywords:
Models, Dyes, Adsorption, Langmuir, Freundlich, AdsorbentAbstract
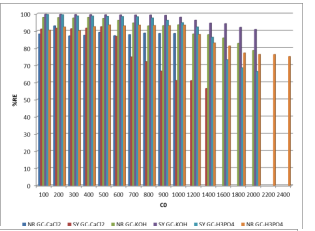
This study aims to evaluate the effectiveness of grapes cores treated by different chemical agents in order to compare their removal efficiency of the anthraquinonic diazo azo (Supranol yellow 4GL) the Azo anionic acid (Nylosan red N-2RBL) dyes from aqueous media. Modeling of isotherms was used to select the best adsorbent; the best adsorption energies, capacities and intensities. Fitted models and data were evaluated by using the most common isotherms models namely Langmuir and Freundlich at their linear and non-linear forms. Parameters and best fit coefficient R2 were identified. Results were statically analyzed by calculating the following errors functions: ?2 statistic; RMSE and API. By using 4 g/L of adsorbent and 100 mg/L as initial NR-dye concentration; the uptakes by GC-2 and GC-3 are 90.5 and 98 % respectively, and 88.31 % onto GC-1 for adsorbent dose of 8 g/L. The adsorption process of NR-dye by GC-3 is very well represented by Freundlich isotherm in its linear and non-linear forms, the adsorption energies of 28.15 and 42.57 mg/g respectively, are the most important, in the other hand, adsorption energy found from the non-linear Langmuir isotherm is very low (0.0203 L/mg). The removal efficiencies of SY-dye by GC-2, GC-3 and GC-1 attained 99.63 %, 99.78 % and 91.9 % respectively at an initial concentration of 100 mg/L and adsorbent dose of 4 g/L. Values of maximum adsorption capacities and energies from different linear and non-linear Langmuir isotherms models for the adsorption of SY-dye by GC-3 were found to be the most important, and the non-linear models were found more suitable. In the other hand, non-linear Freundlich isotherms models are more significant than linear, and the process of the adsorption of SY-dye by GC-3 is more favorable because the adsorption energies and intensities were found very important.
References
M. Hamzaoui, B. Bestani, N. Benderdouche, Z.Mekkibes1, O. Douinat, “Adsorption of Nylosan Red onto Grapes Cores-based Activated carbon”, International Journal of Scientific Research in Chemical Sciences (IJSRCS), Vol.7, Issue.3, pp.11-19,2020.
M. Hamzaoui, B. Bestani, N. Benderdouche, Z.Mekkibes1, O. Douinat, “Anionic dye N-2RBL Removal by grape seeds-based activated carbon: A comparative study”, International Journal of Scientific Research in Chemical Sciences (IJSRCS), Vol.7, Issue.4, pp.13-20,2020.
M. Hamzaoui, B. Bestani, N. Benderdouche, “The use of linear and nonlinear methods for adsorption isotherm optimization of basic green 4-dye onto sawdust-based activated carbon”, Journal of Materials and Environmental Sciences (JMES), Vol.9, Issue.4, pp.1110-1118,2018.
I.Langmuir, “The adsorption of gases on plane surfaces of glass, mica, and platinum,” J.Am.Chem.Soc. Vol.40, pp.1361-1368, 1918.
H.Freundlich, “Adsorption in solution,” Phys.Chem.Soc. Vol.40, pp.1361-1368, 1906.
C.Theivarasu, S.Mylsamy, “Equilibrium and kinetic adsorption studies of Rhodamine-B from aqueous solutions using cocoa (Theobroma cacao) shell as a new adsorbent,” International Journal of Engineering Science and Technology, Vol.2, Issue.11, pp.6284-6292, 2010.
Harrison,Ferrin;Katti,S.K, “Optimal Design in and Hazards of linearization of Langmuir’s Nonlinear Model.” Int. J. Environ. Sci. Tech., Vol.6, Issue. 3, pp.451-456, 2009.
Hoda Roushdy Guendy, “Treatment and Reuse of Wastewater in the Textile Industry by Means of Coagulation and Adsorption Techniques,” Journal of Applied Sciences Research, Vol.6, Issue.8, pp.964-972, 2010.
John U. Kennedy Oubagaranadin, Z.V.P. Murthy and P.S. Rao. “Applicability of Three-parameter Isotherm Models for the Adsorption of Mercury on Fuller’s Earth and Activated Carbon,” INDIAN CHEMICAL ENGINEER, Indian Institute of Chemical Engineers.Vol. 49, Issue. 3, pp.196-204,2007.
Downloads
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution 4.0 International License.
Authors contributing to this journal agree to publish their articles under the Creative Commons Attribution 4.0 International License, allowing third parties to share their work (copy, distribute, transmit) and to adapt it, under the condition that the authors are given credit and that in the event of reuse or distribution, the terms of this license are made clear.





