Phytochemical screening, Anticancer and cytotoxicity of Pulicaria crispa (whole plant)
Keywords:
Pulicaria crispa, Asteraceae, Cytotoxicity, Sulphorhodamine-B, Breast cancer cell, Colon cancer cell, Prostate cancer, Brine ShrimpsAbstract
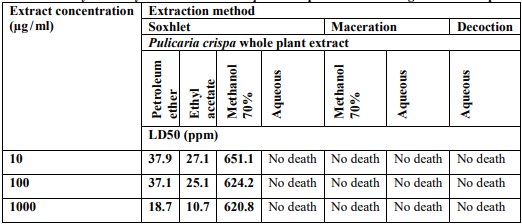
Pulicaria crispa (Asteraceae) is an indigenous plant in Western Sudan. All parts of the plant are traditionally used to heal various diseases, especially those related to the respiratory system. The aim of this study was the chemical screening of metabolites extracted from the plant. Besides, test their toxicity, and their inhibition effect on cancer cells. The metabolites were completely extracted from the plant using three methods: Soxhlet with petroleum ether, ethyl acetate, and ethanol. Maceration with 70% methanol, and decoction with distilled water. The results of chemical screening of all extracts obtained, except the petroleum ether extract, showed the presence of alkaloids, flavonoids, phenols, carbohydrates, terpenes, proteins, and lipids. The Soxhlet petroleum ether extract showed no alkaloids, carbohydrates, and proteins. The cytotoxicity of plant metabolites was tested using brine shrimp lethality assay. The results showed that Soxhlet extracts revealed significant toxicity (P <0.05) to brine shrimp larvae compared to other extracts. Anticancer was investigated by sulforodamine B (SRB) screening against three lines of cancer cells: breast cancer cell line (MCF7), colon cancer cell line (PC3), and prostate cancer cell line (HCT116). The results reflected that Soxhlet extracts had a significant effect on inhibiting the growth of breast and colon cancer cells besides ethyl acetate extract was also inhibit the growth of prostate cancer cells. The maceration and decoction extracts were revealed no anticancer activity against all cancer cells tested.
References
D.J.Newman, G.M. Cragg. Natural products as sources of new drugs from 1981 to 2014. J Nat Prod; 79(3): pp.629-661, 2016.
D.L. Moreira, S.S.Teixeira, M.H.Monteiro, De-Oliveira ACAX, F.J.Paumgartten. Traditional use and safety of herbal medicines. Rev Bras Farmacogn ; 24(2): pp.248-257, 2014.
A.G. Atanasov, B. Waltenberger, E.M. Pferschy-Wenzig, T. Linder, C.Wawrosch, P.Uhrin, et al. Discovery and resupply of pharmacologically active plant-derived natural products: A review. Biotechnol Adv; 33(8): 1582-1614,2015.
A. Zardi-bergaoui et al., “Chemical composition and bioactivities of essential oils from Pulicaria vulgaris subsp. dentata ( Sm.) Batt. growing in Tunisia,” J. Essent. Oil Res., vol. 00, no. 00, pp. 1–10, 2019.
A. Fahmi, N. Hassanen, M. Abdur-Rahman, and E. Shams-Eldin, “Phytochemicals, antioxidant activity and hepatoprotective effect of ginger (Zingiber officinale) on diethylnitrosamine toxicity in rats,” Biomarkers, vol. 24, no. 5, pp. 436–447, 2019.
Y. H. Elshiekh and M. A. M. Abdelmageed, "Antioxidant, Cytotoxicity and Antitumor of Bergia suffruticosa ( whole plant ), J. IJSRBS” vol. 6, no. 6, pp. 52–55, 2019.
A. Esmael et al., “Antimicrobial activity of certain natural-based plant oils against the antibiotic-resistant Acne bacteria,” Saudi J. Biol. Sci., 2019.
S. Pharmacological, A. Of, A. Extracts, and O. F. Some, “International Journal of Innovative Pharmaceutical Sciences and Research,” vol. 3, no. 124, pp. 124–132, 2015.
E. H. M. Ahmed, B. Y. M. Nour, Y. G. Mohammed, and H. s. Khalid, “Antiplasmodial Activity of Some Medicinal Plants Used in Sudanese Folk-medicine,” Environ. Health Insights, vol. 4, no. 1, 2010.
E. D. Benjamin, G. A. Ishaku, F. A. Peingurta, and A. S. Afolabi, “Callus Culture for the Production of Therapeutic Compounds,” vol. 4, no. 4, pp. 76–84, 2019.
M. Elshafei, E. Habiballa, and M. A. Mohamed, “Antibacterial Effects of Quercus Infectoria on Bacteria Isolated from Urinary Tract Infection Patients,” vol. 4, no. 3, 2019.
M. A. Mohamed, “EFFECT OF PROCESSING ON AMINO ACIDS CONTENTS OF FOUR SPECIES OF THE,” vol. 6, no. 9, pp. 3775–3780, 2015.
BIOASSAY TECHNIQUES FOR DRUG.
M. A. Emam, H. I. Khattab, and M. G. A. Hegazy, “Assessment of anticancer activity of Pulicaria undulata on hepatocellular carcinoma HepG2 cell line,” Tumor Biol., vol. 41, no. 10, pp. 1–10, 2019.
E. E. Elgorashi and L. J. Mcgaw, “South African Journal of Botany African plants with in vitro anti-in fl ammatory activities : A review,” South African J. Bot., vol. 126, pp. 142–169, 2019.
A. Activity, A. Of, and E. Extract, “International Journal of Innovative Pharmaceutical Sciences and Research ANTIBACTERIAL ACTIVITY ASSESSMENT OF RHIZOME,” vol. 5, no. 1, pp. 1–11, 2017.
S. S. Shiva, ur da clo edia. .
P. Application and D. D. From, “Biological Assay,” pp. 1–14.
B. Berke, A. Umar, C. Cheze, H. Gin, and N. Moore, “A review of Algerian medicinal plants used in the treatment of diabetes,” J. Ethnopharmacol., vol. 238, no. April, p. 111841, 2019.
O. A. O. Adam, R. S. M. Abadi, and S. M. H. Ayoub, “Effect of Extraction method and Solvents on yield and Antioxidant Activity of Certain Sudanese Medicinal Plant Extracts,” J. Phytopharm., vol. 8, no. 5, pp. 248–252, 2019.
“Phytochemical screening and Antimicrobial activity of seeds and fruits extract of Momordica balsamina Linn,” vol. 20, no. 2, pp. 1–7, 2019.
H. S. Eldeenizzeldeenabdalaziz and M. A. M. Mageed, “Photochemical Analysis, Antioxidant Activity and Cytotoxicity of Adansonia digitata L. Seeds Ethanol Extract,” vol. 1, no. 1, 2019.
Downloads
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution 4.0 International License.
Authors contributing to this journal agree to publish their articles under the Creative Commons Attribution 4.0 International License, allowing third parties to share their work (copy, distribute, transmit) and to adapt it, under the condition that the authors are given credit and that in the event of reuse or distribution, the terms of this license are made clear.





