Synthesis, Characterization and Biological Aspects of O- and N-Donor Schiff Base Ligands and their Co(ll),Cu(ll),and Ni(ll) Complexes
Keywords:
Azomethine, Biological activities, Condensation, DMF, DMSO, Furfuraldehyde, SynonymousAbstract
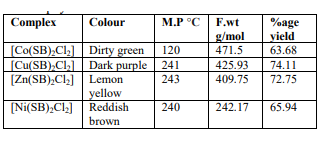
A Schiff base is a compound with the general structure R₂C=NR`. Schiff bases and their complexes are flexible compounds synthesized from the condensation of an amino compound with carbonyl compounds and extensively used for industrial purposes and also show a broad range of biological activities including antibacterial, antifungal, antiviral, antimalarial, antiproliferative, anti-inflammatory, anticancer, anti-HIV, anthelminthic and antipyretic properties. Many Schiff base complexes show excellent catalytic activity in various reactions and in the presence of moisture. They can be considered a sub-class of imines. The term is often synonymous with azomethine which refers specifically to secondary aldimines.These are formed by the condensation reaction of primary ammines and carbonyl compounds. Furfuraldehyde and it’s various derivatives are also useful in the synthesis of Schiff bases. Schiff base forms a stable complex with metals in +2 oxidation state. Keeping all these facts into consideration, Here four new Complexes Co(ll),Zn(ll),Ni(ll) and Cu(ll) with O- and N- donor Schiff bases derived from Furfuraldehyde and Ethylenediamine have been synthesized and Characterized. In this chemical synthesis the ligands of schiff base was synthesized by the reaction of furfuraldehyde with Ethylenediammine in 2:1 ratio respectively in methanol. The analytical results and molecular weights of the compounds support the formulations. The obtained coloured crystalline Complexes are soluble in acetone and DMF but sparingly soluble in DMSO.
References
S. Yamda, Coord. Chem. Rew., 192, 537 (1999).
G. Jian and X. Tong-Tao, Synth. React. Inorg. Met-Org Nano-Met. Chem., 38, 50 (2008).
. W. G. Hanna and M. Moawaad, Trans. Met. Chem., 26(6), 644 (2001).
Bhakti, J. Sci. Ind. Res., 114, 1213 (1953).
Z. Poddar, Ana. Chem., 154, 324 (1957).
Ullman’s Encyclopedia of Ind. Chem., Willey Veh., 26, 614 (1984).
C. H. Collins and Lye, Microbiological Methods, 4th Ed., Butterworth, London, (1976) p. 235.
A. I. Vogel, A Text Book of Quantitative Inorganic Analysis, 3rd Edn., ELBS, London (1978).
S. I. Mostafa, T. H. Rakha and M. M. El Agex, Indian J. Chem., 39(A), 1301 (2000).
R. K. Dubey, A. N. Mishra and C. H. Mishra, Proc. Nat. Acad. Sci., India, 75(A), 239 (2005).
A. P. Mishra and S. K. Gautam, J. Indian Chem. Soc., 81, 324 (2004).
M. Thomas, M. K. M. Niar and P. K. Radhakrishnan, Synth. React. Inorg. Met-Org Chem., 25, 471 (1995).
C. R. Bhattachajee, P. Goswami, S. Neogi and S. Dhibar, Assam Univ. J. Sci. and Tech.; Phy. Sci.and Tech., 5(II), 81 (2010).
W. Kemp, Organic Spectroscopy, ELBS, Hong Kong, (1987).
G. C. Percy and D. A. Throton, J. Inorg. Nucl. Chem., 34, 3357 (1972).
J. T. Makode and A. S. Aswar, Indian J. Chem., 43(A), 2120 (2004).
P. Muthumanim, S. Venkataraman, R. Meera, G. Nayak, N. Chidambaranathan, P. Devi and B. Kameshwari, Der. Pharma Chemica., 2(1), 385 (2010).
N. Raman, A. Kulandaisamy and K. Jayasubramaniam, Indian J. Chem., 41(A), 942 (2002).
Downloads
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution 4.0 International License.
Authors contributing to this journal agree to publish their articles under the Creative Commons Attribution 4.0 International License, allowing third parties to share their work (copy, distribute, transmit) and to adapt it, under the condition that the authors are given credit and that in the event of reuse or distribution, the terms of this license are made clear.





