Separation of a mixture of benzoic acid (C6H5COOH) and benzophenone (C6H5COC6H5) by extraction
Keywords:
Extraction, Separation, Benzoic Acid, Benzophenone, PhenolAbstract
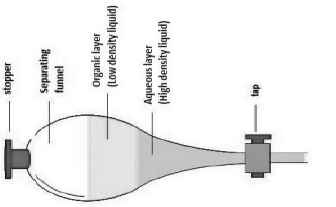
Extraction is a technique used to isolate and purify organic compounds when they are in a mixture with other compounds. The reactions between acids and bases were initially studied, with the solubilization and recovery of both the acidic and the basic properties of the compounds. Benzoic acid, naphthole and acetic acid are the acids that reacted with ΝaOΗ, Na2CO3 and NaHCO3 and the corresponding salt was formed. Naphtol did not react with NaHCO3 because it is a weaker acid. Depending on the ability to react with or not an acid with carbonates, the compound can be identified as carboxylic acid or phenol. Salt solutions were neutralized with HCl and the original insoluble acids were re-formed. This was followed by the dissolution of amine with HCl to form its salt, and the original molecule was recovered with NaOH. In addition, a mixture of benzoic acid and benzophenone was separated by extraction. Dichloromethane was used as the solvent and the solution was dissolved with NaOH. As a result, benzoic acid was moved to the aqueous layer as salt and benzophenone was moved to the organic layer. After these two layers were separated, the salt of the benzoic acid was neutralized with HCl and the initial acid was formed.
References
J.C. Gilbert, S.F. Martin, Experimental Organic Chemistry, a Miniscale and Microscale Approach, Brooks/Cole Publisher, USA, pp. 153-178, 2011
R.B. Thompson, Illustrated Guide to Home Chemistry Experiments, All Lab, No Lecture, O’Reilly Publication, USA, pp. 105-107, 2008
R. Blumberg, Liquid-Liquid Extraction, Academic Press Publisher, USA, pp. 73-92, 1988
L.G. Wade Jr., Organic Chemistry, Pearson Education Publication, USA, pp. 944-948, 2013
J. McMurry, Organic Chemistry, Crete University Press Publisher, Greece, pp. 1140- 1166, 1204-1218, 2012
Downloads
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution 4.0 International License.
Authors contributing to this journal agree to publish their articles under the Creative Commons Attribution 4.0 International License, allowing third parties to share their work (copy, distribute, transmit) and to adapt it, under the condition that the authors are given credit and that in the event of reuse or distribution, the terms of this license are made clear.





