Topological Modeling of log D7.4 of Hydroxylated Aromatic Aldehydes
Keywords:
Toxicity, logD7.4, topological indices, QSAR, regression analysisAbstract
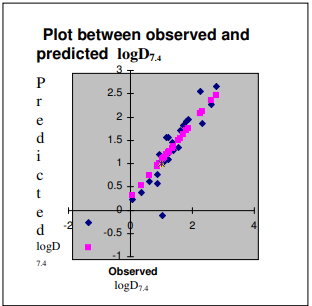
The quantitative Structure-Aciivity Relationship (QSAR) was performed for a set of 25 hydroxylated aromatic aldehydes for modeling logD7.4 , responsible for exhibiting toxicity ,using topological indices . The purpose of the study is, therefore, to find out topological dependence of logD7.4 vis-à-vis toxicity. Multiple regression analysis (MLR)was used for obtaining statistically significant models. The results show that statistically significant models are obtained in multi-parametric regression model in that Ss,Xu and MSD are found useful in modeling of logD7.4 .
References
Anon 2001, White paper on Strategy for a future chemicals policy.
Cronin, M.T.D.; Walker, J.D.; Jaworsha, J.C.; Cumber, M.H.T.; Watts, C.D.; Worth, A.P., Environ. Health Perspect, 2003, 111, 1376-1390.
Priace, R.C.; Gunson, D.E., Treads Biochem. Sci., 1994, 19, 521-530.
Netzeva, T.I.; Schultz, T.W., Chemosphere, 2005, 61, 1632-1643.
Karabunarliev, S.; Mekenyan, O.G.; Karcher, W.; Russom, C.J.; Bradbury, S.P., Quant. Struct. Act. Relat., 1996, 4, 302-310.
McKim, J.M.; Schmieder, P.K.; Niemi, G.J.; Carlon, R.W.; Heary, T.R., Environ. Toxicol. Chem., 1987, 6, 313-328.
Patlewicz, G.; Basketter, D.A.; Smith, C.K.; Hotckkine, S.A.; Roberts, D.W., Contact Derm, 2001, 44, 331-336.
Benigni, R.; Passerini, L.; Redomoate, A., Environ. Mol. Metagen, 2003, 42, 136-143.
DRAGON, Evaluation ,version 5.0 software
CHEM SKETCH 10.0 software
Downloads
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution 4.0 International License.
Authors contributing to this journal agree to publish their articles under the Creative Commons Attribution 4.0 International License, allowing third parties to share their work (copy, distribute, transmit) and to adapt it, under the condition that the authors are given credit and that in the event of reuse or distribution, the terms of this license are made clear.





