Spectrophotometric study of the oxidation of fosfomycin by potassium permanganate in aqueous perchloric acid medium _ a kinetic and mechanistic approach
Keywords:
Fosfomycin, Permanganate, Kinetics and mechanismAbstract
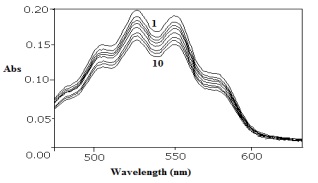
The kinetics of oxidation of fosfomycin (FOS) by potassium permanganate in perchloric acid medium has been studied spectrophotometrically at 25 oC and at constant ionic strength of 3.60 mol dm-3. The stoichiometry of the reaction is determined and found that one mole of permanganate requires one mole of FOS (1:1). The reaction shows first-order dependence in permanganate and apparently less than unit order in both fosfomycin and acid concentrations. The identified oxidation products are Mn2+, formyl phosphonic acid and acetic acid. Reaction rate increases with increase in ionic strength. The increase in the acetic acid content in the reaction medium decreased the rate of the reaction. Based on the experimental results a mechanism involving complex formation between MnO4- and substrate has been proposed. The reaction constants involved in the different steps of mechanism are calculated and activation parameters with respect to thermodynamic quantities are computed and discussed. From this spectroscopic investigation we proposed suitable scheme of the experiment.
References
B.S. Perez, A.L. Sorai, M.O. Tapia, J. Anim. Product. Adv. 2013, 4, 107
P. Espandiari, J. Zhang, B.A. Rosenzweig, Y. Zhou, V.S. Vaidya, L. Schnackenberg, Faseb J. 2008, 22, 917
P. Savignac, B. Iorga, Modern phosphonate chemistry. CRC press, 2003
M. Neuman, G. Fluteau, Intern. J. Experim. Clinic. Chemoth. 1977, 23, 196
R.R. Alexandro, M.D. Maria, C. Alejandro, G. Cristina, C.G. Alfredo, O. Antonio, J. Blazquez. PLoS One, 2010, 5, 10193
K.B. Wiberg, Oxidation in Organic Chemistry, Part A, Academic, New York, 1965, 6, 57
M.C. Day, J. Sebin, Theoretical Inorganic Chemistry. Reinhold Publishing Corporation, New York, 1985, 344
P.N. Naik, S.A. Chimatadar, S.T. Nandibewoor, Ind. Eng. Chem. Res. 48, 2548 (2009)
P. K. Sen, A. Saniyal, K. K. Gupta, Inter. J. Chem. Kin. 1995, 27, 379
A. I. Vogel, Vogel’s text book of macro and semi macro Qualitative inorganic analysis. John Wiley & Sons, New York, 1967, 291
F. Figel, Spot Tests in Organic Analysis. Elsevier, New York. pp 435 (1975)
L. Gabor, F. Istvan, J.P.J.A. Anthony, New J. Chem., 29, 759 (2005)
K.S. Byadagi, RV. Hosahalli, S.T. Nandibewoor, S.A. Chimatadar. Ind. Eng. Chem. Res. 2011, 50, 10962
K. S. Byadagi, R. V. Hosahalli, S. T. Nandibewoor, S.A. Chimatadar, Ind. Eng. Chem. Res. 2011, 50, 10962; N. Bailey, A. Carrington, A.K. Lon, M.C.R. Symons, J. Chem. Soc. 1960, 290
S. Dash, S. Patel, B. K. Mishra, Tetrahedron 2009, 65, 707
S. Pragya, K.U. Santosh, Ind. J. Chem., 2008, 47, 1037
M.H. Refat, Can. J. Chem., 1991, 69, 2018
S.A. Farokhi, S.T. Nandibewoor, Can. J. Chem., 2004, 82, 1372
D.C. Hiremath, K.S. Tyabaj, S.T. Nandibewoor, Int. J. Chem. Kin., 2007, 39, 236
S.J. Malode, J.C. Abbar, S.T. Nandibewoor, Inorg. Chem. Acta., 2010, 363, 2430
Downloads
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution 4.0 International License.
Authors contributing to this journal agree to publish their articles under the Creative Commons Attribution 4.0 International License, allowing third parties to share their work (copy, distribute, transmit) and to adapt it, under the condition that the authors are given credit and that in the event of reuse or distribution, the terms of this license are made clear.





