Development and Validation of Analytical Methods for Pharmaceutical Drug Products
Keywords:
Analytical Methods, Pharmaceutical Drugs,, Method Validation, High-Performance Liquid Chromatography, Paracetamol, Stability StudiesAbstract
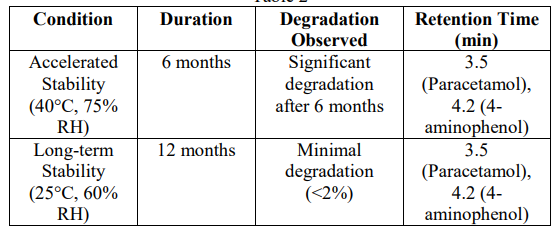
This paper focuses on the development and validation of analytical methods to ensure the quality, efficacy, and safety of pharmaceutical drug products. The study covers various analytical techniques, including High-Performance Liquid Chromatography (HPLC), Ultraviolet-Visible (UV-Vis) Spectroscopy, and Mass Spectrometry (MS), and their application in the quantitative analysis of active pharmaceutical ingredients (APIs). A case study on a model drug, Paracetamol (C8H9NO2), is presented to demonstrate the process of method development and validation. Stability studies were also conducted to evaluate the degradation profile of the drug under different conditions.
References
International Conference on Harmonisation (ICH) Q2(R1), "Validation of Analytical Procedures: Text and Methodology," 2005.
Snyder, L. R., Kirkland, J. J., & Dolan, J. W. "Introduction to Modern Liquid Chromatography," John Wiley & Sons, 2009.
Sharma, B. K. "Instrumental Methods of Chemical Analysis," Goel Publishing House, 2000.
ICH Q1A(R2) Stability Testing of New Drug Substances and Products, International Conference on Harmonisation, 2003.
Huber, L. Validation of Analytical Methods: Review and Strategy, LC-GC Europe, Vol.20, Issue.5, pp.96-101, 2007.
Brittain, H. G. (Ed.). Profiles of Drug Substances, Excipients, and Related Methodology, Vol. 41, Academic Press, 2016.
Jenke, D. R., Chromatographic Method Validation: A Review of Current Practices and Procedures, Journal of Liquid Chromatography & Related Technologies, Vol.37, Issue.3, pp.344-357, 2014.
Snyder, L. R., Kirkland, J. J., & Dolan, J. W. Practical HPLC Method Development, 2nd Edition, John Wiley & Sons, 2010.
United States Pharmacopeia (USP) 42-NF 37, General Chapters: Validation of Compendial Procedures, United States Pharmacopeial Convention, 2019.
Downloads
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution 4.0 International License.
Authors contributing to this journal agree to publish their articles under the Creative Commons Attribution 4.0 International License, allowing third parties to share their work (copy, distribute, transmit) and to adapt it, under the condition that the authors are given credit and that in the event of reuse or distribution, the terms of this license are made clear.





