Toxicity Study and in silico ADME of some selected Anti-HIV agents inhibitors
Keywords:
Anti-HIV agents, Toxicity, Validation, Y- randomizationAbstract
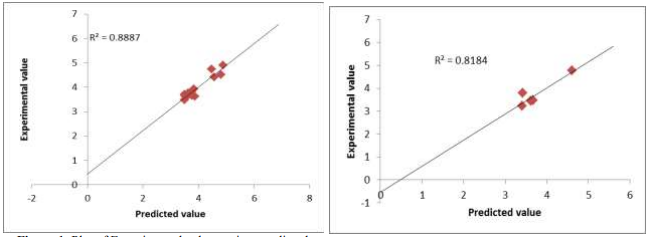
Quantitative structural and cytotoxicity relationship (QSCR) has been carried out on twenty pyrindine-4-carboxylate derivatives using genetic function approximation combined with multiple linear regression (GFA-MLR). The following results were obtained by GFA-MLR using three descriptors AATS6i, MAT57e and TDB9s; R-squared, R2 = 0.888666, Adjusted R-squared, R2adj= 0.855266, cross validated R-squared, LOO-Q2CV = 0.710242, Y-randomization, cR2p = 0.793663 and external prediction, R2prediction = 0.8184. Their physiochemical properties are in agreement with a rule given by Lipinski’s rule and results of molecular weight ? 500 daltons, hydrogen acceptors of ? 10, hydrogen bond donors of ? 5 and octanol water partition coefficient ? 5 were obtained. This research provides an understanding on new HIV drugs with reduced toxicity and greater effectiveness.
References
K. Roy, J. Leonard, “QSAR modelling of HIV-1 reverse transcriptase inhibitor 2-amino-6-arylsulfonylbenzonitriles and congeners using molecular connectivity and t-state parameters, “Bioorg Medicinal. Chemistry, Vol.12, Issue. 4, pp. 745-754, 2004.
A.S.Ugochukwu, , G. A. Shallangwa, A. Uzairu., “Quantum and statistical study for evaluating the cytotoxicity ability of some pyrazole derivatives as potent Anti HIV-1 agents inhibitors, “International journal of New Chemistry, Vol. 8 Issue. 2, pp. 131-141, 2021.
F. Ooms, “Molecular modeling and computer aided drug design. Examples of their applications in medicinal chemistry, “Current medicinal chemistry, Vol. 7 Issue. 2, pp. 141-158, 2000.
G. Liu, R. Luo, X. ZHANG, Y. ZHOU, J. LI, Y. ZHENG, H. Liu, “Synthesis and Evaluation of Anti-HIV Activities of Novel 7-Hydroxy-1,3-dioxo-2,3-dihydro-1H-pyrrolo[3,4-c]pyridine-4-carboxylate Derivatives, “Medicinal chemistry, Vol.4 Issue. 8, pp. 573-580, 2014.
S.U. Ahanonu, S. U, G. A. Shallangwa, U. Adamu, “Quantitative structure and activity relationship of 3a, 6a – Dihydro-1H- pyrrolo[3,4-c] pyrazole-4,6-dione “Turkish Computational and Theoretical Chemistry, Vol. 4, Issue. 1, pp. 32 – 38, 2020.
A. Tropsha, “Best practices for QSAR model Development, Validation and Explitation, “Mol. Inf, Vol. 29, Issue. 6, pp. 476-488, 2010.
K. Roy, “Some aspects of validation of predictive quantitative structure-activity relationship models, “Expert Opinion On Drug Discovery, Vol. 2, Issue. 12, pp. 1567-1577, 2007.
S. A. Asma, A. S. Hassan, A. M. Naglah, “In vitro cytotoxicity and druglikeness of pyrazolines and pyrindines bearing benzofuran moiety, “Journal of Applied pharmaceutical science, Vol. 10, Issue. 4, pp. 142-148, 2020.
C. A. Lipinski, F. Lombardo, B. W. Dominy, P. J.Feeney, “Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings, “Journal of Advanced Drug Delivery. Reviews, Vol. 46 Issue. 1, pp. 3-26, 2001.
Downloads
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution 4.0 International License.
Authors contributing to this journal agree to publish their articles under the Creative Commons Attribution 4.0 International License, allowing third parties to share their work (copy, distribute, transmit) and to adapt it, under the condition that the authors are given credit and that in the event of reuse or distribution, the terms of this license are made clear.





